Content Overview
This article focuses on the use of a wellbore cement additive:
Gilsonite as a potential agent in the mitigation of micro-annular flow through its unique affinity to hydrocarbons. Gilsonite’s affinity and potential to absorb hydrocarbons in the cement matrix is analyzed thoroughly through microstructural analysis of hydrocarbon interaction with Gilsonite grains. The goal of this project is to provide a proof of concept for further microstructural analysis of Gilsonite’s ability to mitigate microannular flow. To accomplish this, experimentation was broken into two sperate phases. Phase 1 consists of experimentation on pure Gilsonite to gauge if there was an affinity between Gilsonite and hydrocarbons, while Phase 2 consists of experimentation on Gilsonite rich cement.
The innovative use of Gilsonite as a wellbore cement additive presents a promising solution to the pervasive issue of micro-annular flow in the oil and gas industry. Micro-annular flow, the unwanted movement of gases or fluids in the annular space between the casing and the wellbore, can lead to significant operational and environmental challenges. Gilsonite, a naturally occurring solid hydrocarbon, is being explored for its unique ability to interact with other hydrocarbons, potentially serving as a barrier to this flow. The initial phase of this research project delved into the intrinsic properties of Gilsonite, particularly focusing on its hydrocarbon affinity. Through rigorous testing, the aim was to determine whether Gilsonite inherently possesses the ability to absorb or interact with hydrocarbons. This phase involved a detailed microstructural analysis, observing how Gilsonite grains respond to the presence of hydrocarbons and whether this interaction could be harnessed effectively in wellbore cement formulations.
Advancing to Phase 2, the research expanded to incorporate Gilsonite within cement mixtures used in wellbore applications. This phase aimed to understand how the presence of Gilsonite affects the overall properties of the cement, especially its resistance to micro-annular flow. Experimentation in this stage involved creating various Gilsonite-rich cement samples and subjecting them to conditions that simulate real-world wellbore environments. The focus was to observe how the Gilsonite-modified cement reacts to hydrocarbons and whether its integration could enhance the cement’s ability to prevent micro-annular flow. This comprehensive analysis included studying the cement’s mechanical strength, permeability, and hydrocarbon absorption capabilities. The outcome of this phase is expected to provide crucial insights into the feasibility of using Gilsonite as a cement additive in wellbore applications. The overarching goal of this two-phased project is to establish a proof of concept for Gilsonite’s effectiveness in enhancing the integrity of wellbore cement, paving the way for safer and more efficient drilling operations in the oil and gas industry.
Introduction
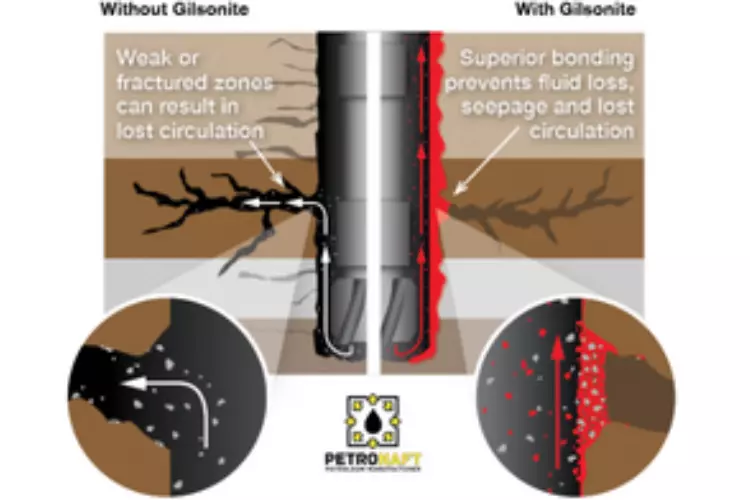
Gas intrusion, known as micro-annular flow, through the cement filled annulus has the potential to create pathways for formation fluids to flow through the cement sheath between formations or to the surface. This unwanted migration of fluids is not only dangerous but can be costly to operators by negatively affecting drilling and production conditions with potential to cause a total blowout (Dusseault et al. 2000). Pathways for fluid to migrate through the well into the annulus pressurizes the well rendering it unsafe for production, which can ultimately cause major safety and economic issues for the operator (Kupresan et al. 2014). This failure could also result in premature water production which can limit the economic life of the well. Remedial cement jobs can in some cases resolve this issue, but at the cost of more time which equates to higher costs thus decreasing total monetary profit. Novel technologies, such as mechanical manipulation of the casing are being evaluated as a faster option (Radonjic & Kupresan, 2015). Mitigation to gas intrusion could be achieved using self-healing cement additives and drilling products.
Self-healing cement additives have the potential to avoid remedial cementing or plug and abandonment of the wellbore by automatically sealing annular pathways after being exposed to unwanted hydrocarbons (Langley et al. 2018). The additives remain dormant in the cement sheath until hydrocarbon migration occurs. Once exposed to hydrocarbons a reaction occurs, expanding the cement and re-establishing an annular seal preventing any future hydrocarbon migration (Le Roy-Delage et al 2010). The focus of this study is a self-healing cement additive known as Gilsonite.
The fundamental objective of cementing is to ensure total zonal isolation between distinct forms and the surface (Bensted & Barnes, 2002).
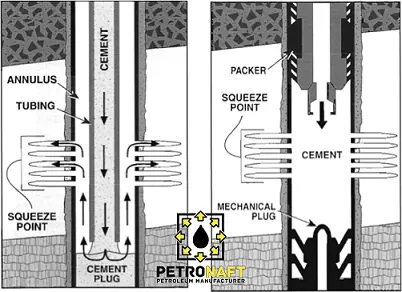
The main purpose of cementing is to provide complete zonal isolation between different formations and the surface, as Portland cement hydration produces material with very low permeability (Bensted & Barnes, 2002). Gas intrusion into the cement filled annulus can lead to the development of pathways in which formation fluids can flow within the cement to either neighboring formations or the surface. This flow can occur in the casing string, or in liners where gas can emerge at the liner hanger which leads to its circulation out of the well. Remediation of this problem can be time consuming and costly, requiring remedial cement measures such as squeeze cementing until there is no more observed migration of unwanted hydrocarbons. Cement squeezes are difficult to perform correctly with a low rate of success.
Self-healing cement (SHC) is a responsive material, where the self-repair mechanism is triggered when hydrocarbons make contact with this material. The main proponent of this technology is that it is self-activated, once exposed to hydrocarbons expansion occurs sealing off areas that were previously exposed to micro-annular flow. Self-healing cements can automatically repair when micro annulus or internal cement cracks are created, thus, repairing and maintaining the integrity of the cement sheath thus allowing it to maintain its functionality. SHC’s provide long term durability to a cement sheath repairing cement sheath integrity without the need of manual well intervention. In addition, SHC act just as regular cement if there is no exposure to hydrocarbons. Reduced costs of cement remediation and lost circulation loss in cement integrity while also the potential to improve productive life of the make using SHC an economical and time efficient decision if applicable. (Langley et al. 2018).
It is important to note that the concept of SHC does not preclude the importance of good cement practice, but rather improves the ability to mitigate micro annular fluid migration. When choosing a SHC it is imperative that machinal properties and self-healing properties are both optimized in order to maintain the highest integrity for the cement sheath. Modern completion methods can expose the cement sheath to extreme pressure and temperature cycles and the related stress associated with casing expansion and contraction, incurring damage to the cement structure down hole (Dusseault et al. 2000. The industries perspective has evolved due to the influx of more advanced drilling which uses uniaxial compressive strength to qualify the strength of a cement system. A cement that can withstand substantial pressure and temperature change is key when preparing a slurry to be placed downhole.
1957 was the first documented use of Gilsonite as a cement additive, primarily used in lost circulation zones as lost circulation material due to Gilsonite’s low specific gravity of 1.07.
(Boden, 2014). Gilsonite’s particle size distribution, impermeability, excellent bridging properties, corrosive and acidic resistance, and low water requirements produce a slurry that has high compressive strengths compared to other slurries with similar weights. Although Gilsonite is a lightweight material it is not porous. That coupled with its impermeability, Gilsonite rich cement is protected against premature slurry dehydration. Additionally, Gilsonite can withstand bottom hole temperatures greater than 350 ˚F.
A 1959 study conducted by Slagle and Carter sought to compare the specific properties of Gilsonite rich cement to other slurries with different additives such as diatomic earth materials, bentonite, expanded perlite at equal densities (Slagle & Carter, 1959). Of these studies, Gilsonite performed highest in compressive strength testing under conditions of 100 -140 ˚F and up to 3000 psi. Slagle and Carter were also able to confirm that Gilsonite samples under atmospheric and 3000 psi showed no measurable difference, thus showing there is no void space in Gilsonite.
High compressive strength and the ability to maintain integrity at elevated temperatures make Gilsonite an ideal candidate for not only a cement additive but also a component of a SHC (Didier et al, 2017). Most SHC are only considered to be a short-term solution and most effective while in slurry form. A SHC with an additive such as Gilsonite may have more longterm durability as an agent to mitigate micro-annular flow well after the cement job is finished.
Methods
The experimental procedure for this research can be broken down into two phases. Phase 1 consists of experimentation on pure Gilsonite to gauge if there was an affinity between Gilsonite and hydrocarbons, while Phase 2 consists of experimentation on Gilsonite rich cement. A detailed table of cement slurry compositions can be found in table 3 in Appendix A. Following Phase 1 and 2 analysis, a conclusion and recommendation for further research will be discussed.
Phase 1: Pure Gilsonite was finely ground using a mortar and pestle before being subjected to micro-structural analysis. Fourier Transform Infrared (FTIR) spectra were collected with a Nicolet iS50 FT-IR Spectrometer equipped with two research grade infrared spectrometers. FTIR was used to examine if there was any CO2 absorption by Gilsonite at pressures up to 400 psi. FTIR provides an infrared spectrum of absorption in either a solid, liquid or gaseous material by collecting high-spectral resolution data over wide spectral ranges. Thermal Gravimetric Analysis (TGA) was used to measure mass change over time under constant exposure to CO2 for two hours at a constant temperature of 86 ˚F and then repeated at 122 ˚F.
Phase 2 of this research was conducted on Gilsonite rich cement cores containing varying amounts of Gilsonite ranging from 2.5,5 and 7.5% Gilsonite by weight of cement. All cement slurries were made to be 13.2 ppg to ensure consistency during testing. All cement used in this research was Haliburton Class H cement mixed in a Waring Laboratory blender per API recommended procedure (API 2013). Mixing was conducted using a 4-liter, 3.75 horsepower Waring laboratory blender. Initially, bentonite was mixed with deionized water at 16,000 rpm for 5 minutes. After, Class H cement and Gilsonite were added and mixed for an additional 35 seconds at 20,000 rpm (for neat cement Gilsonite was not added). Following mixing, cement slurries were poured in 3×1 inch cylindrical brass molds. Samples were kept as atmospheric conditions for 24 hours before being removed from molds and submersed in a solution of deionized water and calcium hydroxide to a pH of ~13. The cement cores in the deionized water and calcium hydroxide solution were the placed in an environmental chamber kept at constant temperature, 70⁰C (158⁰F), and humidity of 100% for 21 days. After this time the samples were removed and place in a 0% humidity oven at a constant temperature of 70⁰C (158⁰F) for 24 hours to completely dry the samples in preparation for testing.
Helium-gas Porosimetry was conducted on each core before and after exposure to CH4. An Ultragrain GrainVolume UGV-200 porosimeter was used to collect porosity measurements. The UGV-200 incorporates Boyle’s Law helium gas expansion porosimetry in which the core is placed in a known volume with a known pressure then exposed to 10 cc of helium gas.
SEM images with EDS analysis were created using a FEI Quanta 3D FEG/SEM with dualbeam focused ion beam microscopy. SEM imaging was completed at 20 KV and 27 pA at 3 different magnifications for both nonexposed and exposed 7.5% Gilsonite BWOC. These magnifications range from 100x – 3500x. SEM imaging provides high resolution images of the Gilsonite interaction in the cement structure before and after exposure to CH4. SEM sends high focused energy electron beams at the surface of the sample. Interactions between the electrons and the sample create backscatter electrons and X-rays to display topography and composition.
Cores exposed to CH4 for 24 hours using a flow through device. Cores were placed inside a 1 inch in diameter rubber sleeve that had a thickness of 1.75 inches. The rubber sleeve was then put in a 1.75 inch in diameter rubber sleeve with thickness of .25 inches sealed the 24 inches in length plastic housing forcing CH4 to flow through cement cores.CH4 was flowed through the cores and into a graduated cylinder filled with H2O to dissolve in water.
Results
Fourier Transform Infrared Analysis (FTIR)
Fourier transform infrared spectroscopy is a formidable tool in creating a comprehensive view of the composition and structure/ composition of materials [10]. Alternatively, FTIR can be utilized to observe the absorbance of a material under a given pressure.
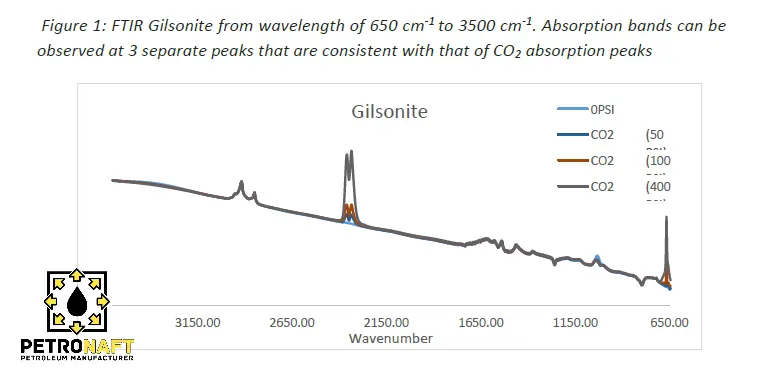
As shown in figure 1 Gilsonite was exposed to C02 under pressures varying from 0 psi to 1200 psi (0,50,100,400, and 1200 psi) with the x axis representing wavelength [cm-1] and the y axis showing absorbance. It should be noted that 1200 psi was not used in results due to poor experimental conditions and leakage observed at that respective pressure. According to the National Institute of Standards and Technology (NIST) for CO2 absorption, if any, will be observed in two major absorption bands. These two bands can be observed in figure 6 in Appendix A. The range of the bands are as follows: 600 cm-1 and 745 cm-1, 2250 cm-1 and 2400 cm-1. FTIR spectra on Gilsonite at all pressures showed 3 major absorption bands. The first band can be observed at 668.22 cm-1, the second band between the range of 2287 cm-1 and 2391.34 cm-1 and the third band between 2786.68 cm-1 and 2991.10 cm-1.
The broad absorption band between 2287 cm-1 and 2391.34 cm-1 is the most conclusive evidence that Gilsonite was readily absorbing CO2. It is important to note that this absorption was observed at 0 psi meaning that at ambient pressure Gilsonite was absorbing CO2 which, strongly suggests that Gilsonite does have an affinity to hydrocarbons. The Absorption band at 2786.68 cm-1 and 2991.10 cm-1 suggests that C-H stretching of O-rings is occurring due to band peaks observed at these wavelengths. The FTIR data collected closely represents the FTIR data from the National Institute of Standards and Technology providing conclusive evidence that Gilsonite is readily absorbing CO2. Understanding that there is absorption between Gilsonite and CO2 is key in moving forward with the viability of Gilsonite as a SHC additive and mitigation tool against microannular flow.
It can be observed that a larger amount of absorption is seen as the pressure increases. While the increase in absorption is likely caused by the increase in pressure, it is common to have increased pressure on the cement in wells with sustained casing pressure. A previous study on sustained casing pressure in 26 different wells measured the frequency of sustained casing pressure. Results of this study showed pressure reaching 1000 psi in production and intermediate casing while reaching 500 psi in surface and conductor casing. While it is evident that Gilsonite does absorb CO2 with and without pressure, sustained casing pressure could potentially contribute to the absorption of hydrocarbons in Gilsonite.
Thermal Gravimetric Analysis (TGA)
TGA results, seen in Figures 2A and 2B below, showed an increase in the weight of Gilsonite particles after exposure to CO2 at constant temperatures of 86 ˚F and 122 ˚F. The weight change is measured as weight% which can by dividing the weight at any time by the initial weight measured prior to running the test. At 86˚F Gilsonite showed a 0.2454% weight increase under constant exposure to CO2 while and a .1956% increase at 122 ˚F. The beginning of each run began with a 5 minute “purge” cycle of nitrogen injection. The loss in weight % during this cycle is most likely attributed to the loss of water or dust mass that was on the surface of the Gilsonite particles. The change in weight will be measured from the weight recorded at the end of the “purge” phase to the max peak of the CO2 injection phase.
There is an evident increase in weight % at both temperatures during CO2 exposure. In the 86 ˚F test the weight reached its maximum peak at the 79.10 minute. While in the 122 ˚F test the weight reached a maximum peak at the 86.96 minute. The 122 ˚F test took longer to reach a lower maximum weight than in the 86 ˚F which is most likely due to the increase in temperature which could evaporate any impurities in the CO2 that did not at lower temperatures.
While the 86 ˚F did reach a higher maximum weight, the difference is negligible and the increase in weight % for both temperatures can be viewed as the same.
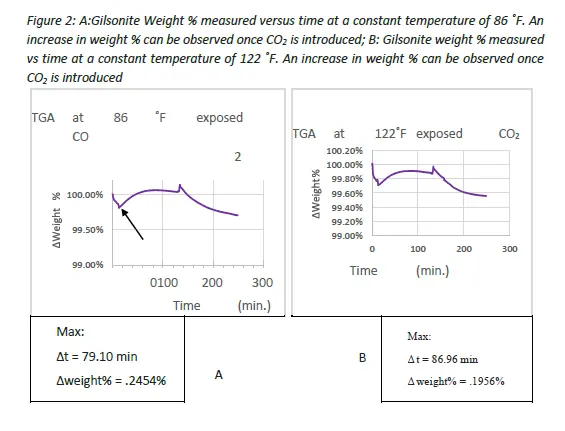
TGA was conducted on samples with a mass of 15.5 and 15.8 micrograms with a mass increase of 0.2454% and .1956% for 86˚F and 122˚F respectively. If these samples were conducted on larger mass samples, as they would be in cement cores it is expected that a proportional increase in weight percentage would increase due to an increase in surface area for absorption. TGA testing of Gilsonite under CO2 injection provides a quantitative analysis that Gilsonite does absorb CO2 at ambient and increased temperatures through weight measurement. TGA coupled with FTIR results both show that Gilsonite readily absorbs CO2 under both pressure and temperature. Since absorption has been observed in two different methods, the next step in testing Gilsonite’s viability as a SHC additive Is understanding the internal structure and then eventually the interaction of Gilsonite in cement.
Porosimetry
Porosity measurements were conducted on samples before and after exposure to CH4. It should be noted that these values were calculated with an error margin of +/- 2.5%. Helium-gas porosimetry provides absolute porosity values rather than effective porosity values. Typical cement slurries with a high water-cement ration (>0.7) have high porosity values above 40% [12]. This corresponds with the data collected on samples with no CH4 exposure. Porosity values decrease with an increase in concentration of Gilsonite. This is evident for both nonexposed and exposed samples. However, the margin of decrease after exposure is much higher for cement that contains Gilsonite compared to neat cement. Gilsonite samples showed a percent decrease of 10.86% for 2.5% Gilsonite, 16.44% decrease for 5% Gilsonite and 14.86% decrease for 7.5% Gilsonite. These decreases are compared to the 4.74% decrease in neat cement containing no Gilsonite.
The decrease in absolute porosity is substantially higher in samples that contained Gilsonite, most notably 5% Gilsonite. This data shows that the absorption shown in FTIR and TGA analysis has a significant impact on the physical properties of cement. A decrease in porosity upon exposure to hydrocarbons with help prevents further flow of those hydrocarbons to surrounding formations or the surface. This porosity data is important to consider when determining the viability of Gilsonite as a SHC additive. Porosimetry provided quantitative analysis that there is an effect on the physical properties of cement post exposure to hydrocarbons.
Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS)
To observe the effects of CH4 exposure on Gilsonite in the microstructure of cement Scanning Electron microscopy (SEM) and Energy dispersive spectroscopy (EDS) were conducted on Gilsonite cores containing 7.5% Gilsonite by weight of cement. SEM Images of Gilsonite in cement before and after exposure were taken at a high, medium and low magnification for comparison. Figure 3 shows exposed and non-exposed samples of 7.5% Gilsonite at a magnification of 100x. At this magnification both the exposed and non-exposed samples, Gilsonite is shown in black and evenly distributed throughout the cement matrix with varying size and shape.

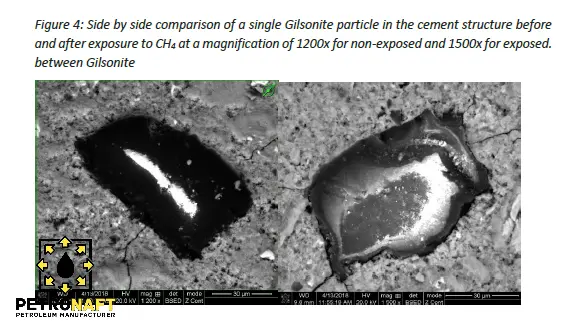
Figure 4 shows the side by side comparison of a single Gilsonite particle in the cement structure before and after exposure to CH4 at a magnification of 1200x for non-exposed and 1500x for exposed. At this magnification it can be observed that in both images there is no chemical reaction present between the cement and the Gilsonite. This is notable in that the relationship between the Gilsonite pieces and cement is only a physical interaction. Fracturing in the cement can be observed in both cases. These cracks stop at the Gilsonite pieces which is a sign that Gilsonite maintains its structural integrity when cracking in the cement occurs, which can increase overall structural integrity of the cement. At the cement/Gilsonite interface the non-exposed sample seems to have more void space at the cement/Gilsonite interface than the exposed sample in which the Gilsonite particles seems to be flush with minimal void space.
Conclusions and recommendations
• Fourier Transform Infrared (FTIR) showed absorption of CO2 at pressures ranging from ambient to 400 psi
• Thermal Gravimetric analysis (TGA) showed CO2 Absorption at 86 ˚F and 122 ˚F
• Porosity measurements showed decrease in absolute porosity with increasing concentrations of Gilsonite with further decrease in porosity after exposure to CH4
• SEM images show no chemical reaction between cement matrix and Gilsonite. Void space less evident in cores exposed to CH4.
Proof of Gilsonite’s potential applicability as a SHC additive can be suggested by the findings of this study. Current remediation methods of microannular flow can be time consuming, which in turn equates to higher costs, dangerous if not detected in time, and not completely effective. Making Gilsonite a logical, cost, efficient, time efficient and a safe alternative to mitigation of microannular flow. Planning and selecting a true SHC is the best defense against micro-annular flow and Gilsonite provides long term stability for wellbore integrity. Further research needs to be completed to make more confident and definitive claims on Gilsonite’s absorption of hydrocarbons and as an agent against microannular flow. The recommendations from this project are detailed in the next section.
To better understand the absorption of hydrocarbons by Gilsonite it is recommended that these same experiments be conducted on a larger time scale, with periods of non-exposure to understand the longevity of this absorption. If long term absorption can be determined, it is recommended that, if possible, a cement/casing and cement/formation interface with micro annuli be produced to scale and exposed to hydrocarbon migration. Another recommendation is to manipulate the Gilsonite grains to all be of the same size and shape with known surface area and then test exposure. If all grains are of equivalent size expansion and absorption on a larger scale can be more easily approximated.
Purchasing This Product from Petro Naft
For more detailed information and to purchase the product discussed in this article, please visit the dedicated product page below. Alternatively, use the various communication channels provided on our site to register your purchase inquiry or take advantage of our expert guidance.
From diverse scientific sources, compiled by the research team of PetroNaft Co.

2 Responses
Dear all, I’ve heard about Gilsonite being used in oilfield applications, particularly related to wellbores. How exactly does Gilsonite help in resolving the issue of leaky wellbores?
Hello and greetings! Gilsonite, in the context of oilfield applications, is often utilized as an additive in drilling fluids. The key factor here is that it has a unique ability to provide a sealing effect in the wellbore. When a wellbore is leaky, it can allow drilling fluids to escape into surrounding formations, which can cause a whole host of problems, from loss of drilling fluids to contamination of groundwater.
By integrating Gilsonite into the drilling fluid, it creates a kind of plugging effect. The small, fine particles of Gilsonite can penetrate into the leaky zones of the wellbore, effectively plugging them up. The Gilsonite then hardens, forming a seal that can withstand the pressures encountered in the wellbore. So in essence, Gilsonite can be considered a solution to leaky wellbores due to its sealing and plugging properties.
However, it is always essential to consider each specific scenario and the complete wellbore system before deciding on the most suitable solution. Consulting with a wellbore expert or a Gilsonite supplier can provide more tailored advice.